Abstract
Despite BCR-ABL-targeting tyrosine kinase inhibitors (TKIs) have dramatically improved the management of chronic myeloid leukemia (CML) patients, TKIs fail to eradicate leukemic stem cells (LSCs). CML-LSCs are recognized as BCR-ABL-kinase activity independent and responsible for TKI-resistance and relapse upon treatment discontinuation. Systematic mapping of druggable signaling pathways enables the identification of novel CML-LSCs effectively targeting drugs, which is critical to enhance the achievement of sustained treatment free remission in CML patients.
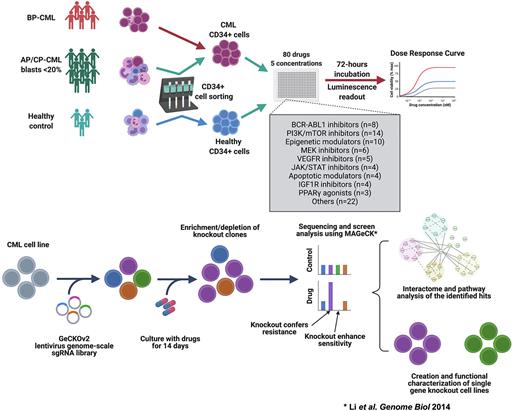
We implemented ex-vivo drug testing of 80 targeted drugs and 18 drug combinations across 10 000-fold concentration, to evaluate drug sensitivity profiles of primary CML-LSCs and progenitors (CD34+ cells) from twenty patients in addition to CD34+ cells from three healthy donors. We identified novel drug vulnerabilities of CML-LSCs including VEGFR-, CDK2/WEE-, MDM2-, and PI3K/mTOR- inhibitors. Combination of imatinib with idasanutlin, venetoclax, AZD1775 and dexamethasone led to synergistic inhibition of CML-LSCs. To delineate the phenotype-specific sensitivity profile of CML CD34+ cells, we applied ex-vivo flowcytometry-based drug screening approach to bone marrow samples from ten CML patients using a custom library of 20 drugs. We identified specific sensitivity of CML CD34+CD38- cells to mepacrine, RG7112, idasanutlin, and navitoclax. In particular, mepacrine showed striking ability to induce differentiation of CML-LSCs, which was further validated in a separate cohort of primary CML samples and cell lines.
To characterize the mechanisms of action of the discovered drugs, we applied genome-scale CRISPR screening in CML cells to identify potential sensitizing and resistance targets for five selected drugs. Imatinib, dasatinib, and tivozanib showed a similar profile of resistance and sensitizing genes, while AZD1775 and mepacrine had unique profiles. We identified knockout targets that confer BCR-ABL1-independent resistance common to different TKIs including mediator complex (MED23, MED26, CCNC), and apoptosis related (APAF1) genes in addition to other genes with uncharacterized role in CML, such as KCTD5 and EIF2AK1. On the other hand, knockout of certain genes was associated with increased sensitivity to specific drugs such as IGF1R-dasatinib and SPNS1-mepacrine associations.
KCTD5 was consistently identified as the top resistance gene to all tested TKIs, and therefore we next investigated the potential role of KCTD5 in CML pathogenesis and TKI resistance. In RNA sequencing data, KCTD5 was significantly downregulated in CML-CD34+ cells compared to healthy counterparts. Using co-immunoprecipitation, we revealed that KCTD5 directly interacts with BCR-ABL and mediate TKI-induced ubiquitination by E3 ligase, as shown by WB assay, that can destine BCR-ABL for subsequent degradation. Knockout of KCTD5 was associated with decreased ubiquitination of BCR-ABL and resistance to imatinib in CML cell lines and primary CML-CD34+ cells. Furthermore, combination of TKI with deubiquitinase inhibitors induced enhanced TKI sensitivity in KCTD5-KO cells.
In conclusion, we discovered novel mechanisms of primary TKI-resistance and drug vulnerabilities of CML-LSCs, which potentially enable eradication of LSCs in CML patients and hence pave the way for the achievement of functional cure in CML patients.
Disclosures
Porkka:AbbVie: Honoraria; Astellas: Honoraria; Bristol-Myers Squibb: Honoraria; Incyte: Honoraria; Novartis: Honoraria; Pfizer: Honoraria; Celgene/Bristol-Myers Squibb: Research Funding; Incyte: Research Funding; Pfizer: Research Funding; Novartis: Research Funding. Mustjoki:Pfizer: Honoraria, Research Funding; Novartis: Honoraria, Research Funding; Bristol-Myers Squibb: Honoraria, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal